Company Profile
Jyong Biotech Ltd. is a science-driven biotechnology company based in Taiwan and are committed to developing and commercializing innovative and differentiated new drugs (plant-derived) mainly specializing in the treatment of urinary system diseases, with an initial focus on the markets of the U.S., the EU and Asia (primarily Taiwan and mainland China).
Since our inception in 2002, we have been dedicated to the research and development of new drugs with high safety and efficacy. Through over 20 years of efforts, we have built integrated capabilities that encompass all key functionalities of drug development, including early-stage drug discovery and development, clinical trials, regulatory affairs, manufacturing and commercialization. Leveraging our strong research and development capabilities and proprietary platform, we have been developing a series of drug candidates. We have resubmitted a new drug application, or NDA, for MCS-2 with the U.S. FDA on December 17, 2021 using Active Pharmaceutical Ingredient (API-1). One of our clinical-stage key drug candidates, PCP, is under phase II trials stage (API-1) in Taiwan. Another preclinical-stage key drug candidate, IC, is under preclinical studies.
The following chart illustrates and summarizes the development status of the pipeline of our drug candidates as of the date of this prospectus:
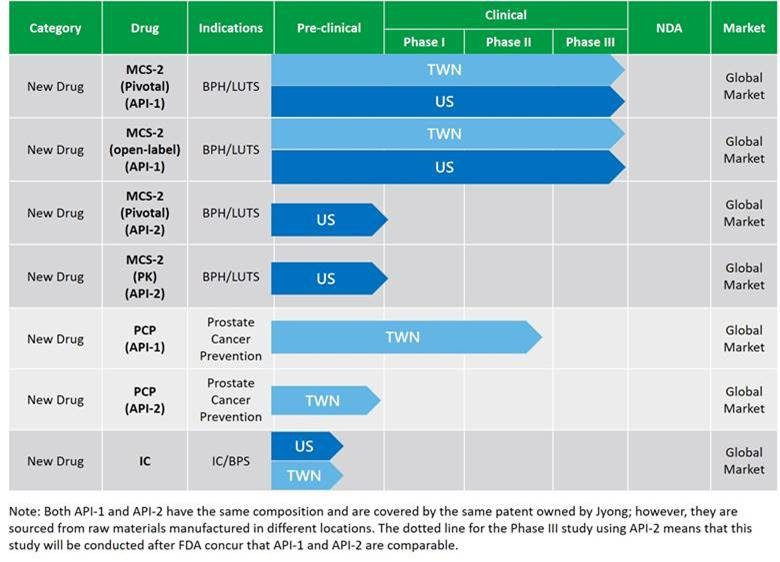
Our pipeline features three innovative and differentiated new drug candidates, and we are developing them for (i) the treatment of benign prostate hyperplasia/lower urinary tract symptoms, or BPH/LUTS, (ii) prostate cancer prevention, and (iii) the treatment of interstitial cystitis, respectively.
MCS-2:
MCS-2 is our new drug candidate developed for unmet medical needs of BPH/LUTS treatment. MCS-2 is expected to be our core product in the future. MCS-2 is a softgel containing patented active pharmaceutical ingredients of carotenoid chylomicrons. The active constituents in MCS-2 are carotenoids, the essential and lipid-soluble component of photosynthetic organisms extracted from plants with eminent photoprotective and antioxidant properties, and we have further developed MCS-2 formulation using chylomicron technology to improve the bioavailability in MCS-2. Chylomicron is a type of lipoprotein particles consisting of triglycerides and phospholipids in human body. MCS-2 has an antioxidant capacity and can reduce inflammatory cytokines. It takes effect by reducing oxidative stress and inflammation and has a dose-response effect. We have completed the four phase III clinical trials for MCS-2 (API-1) in the U.S. and Taiwan, including two pivotal trials and two open-label extension study trials. The pooled study results of our phase III clinical trials on MCS-2 show that the primary endpoint (the change in I-PSS) has reached statistical significance and exhibited safety profile. The incidence of AEs in the MCS-2 group were lower than that in the Placebo group. All SAEs were not-related with MCS-2. BPH/LUTS is the most common urinary tract disease in the middle-aged male population. According to Frost & Sullivan, the global prevalence of BPH increased from 88.4 million in 2017 to 94.2 million in 2020, representing an increase of 6.5%. The global BPH drugs market increased from US$3.7 billion in 2017 to US$4.1 billion in 2020, representing a CAGR of 4.6%. We will set up internal sales and marketing departments, and plan to work with both domestic and international business partners to seize the great market opportunities and to help more patients reduce their distress caused by BPH/LUTS and drug side effects caused by chemical drugs.
Common chemical drugs used to treat BPH include α1-blockers or 5-alpha reductase inhibitors. However,
- 5α-reductase Inhibitors might increase the risk of a more serious form of prostate cancer. [source: US FDA Drug Safety Communication: Safety Announcement June 9, 2011]
- The studies show that men with a benign prostatic hyperplasia diagnosis and exposed to both 5-alpha reductase inhibitor and a-blocker therapy had an increased association with cardiac failure. [source: The Journal Of Urology, May, 2021]
PCP:
PCP is our key new drug candidate developed for the prevention of prostate cancer. (1)Similar to MCS-2, PCP works through its mechanism of antioxidant and anti-inflammatory. PCP contains several types of patented active pharmaceutical ingredients of carotenoid with chylomicron technology that reduce oxidative stress and inflammatory cytokines (IL-6), both of which are main causes of many chronic inflammatory diseases. The active constituents in PCP are carotenoids. PCP and MCS-2 are essentially the same in terms of composition of active ingredients, dosage form, strength and route of administration; however, they are different drug candidates targeting different indications. We are conducting phase II clinical trials of PCP (API-1) in Taiwan. Prostate cancer begins when cells in the prostate gland start to grow out of control. In general, the more quickly prostate cells grow and divide, the more chances there are for mutations to occur. According to Frost & Sullivan, the global prevalence of prostate cancer increased from 10.0 million in 2017 to 11.2 million in 2020, representing a CAGR of 3.9%. The global prostate cancer market increased from US$9.7 billion in 2017 to US$12.6 billion in 2019, representing a CAGR of 9.1%. In addition, the prostate-specific antigen abnormal population, or PSA abnormal population, representing men over 40 years old with a prostate-specific antigen test value of 4.0 ng/ml or higher, is exposed to a high risk of prostate cancer. From 2015 to 2020, the total number of PSA abnormal populations in the U.S., Taiwan and China increased from 5.0 million to 5.3 million.
IC:
IC is our additional key new drug candidate which is composed of polysorbate loaded micelles as nanocarriers which can be used in the intravenous injection and intravesical instillation. The micelles enhance the bioavailability by prolonging the duration of stay in the bladder and increase the penetration of drug across the bladder wall. IC/BPS, refers to a bladder pain disorder that is often associated with voiding symptomatology and other systemic chronic pain disorders.